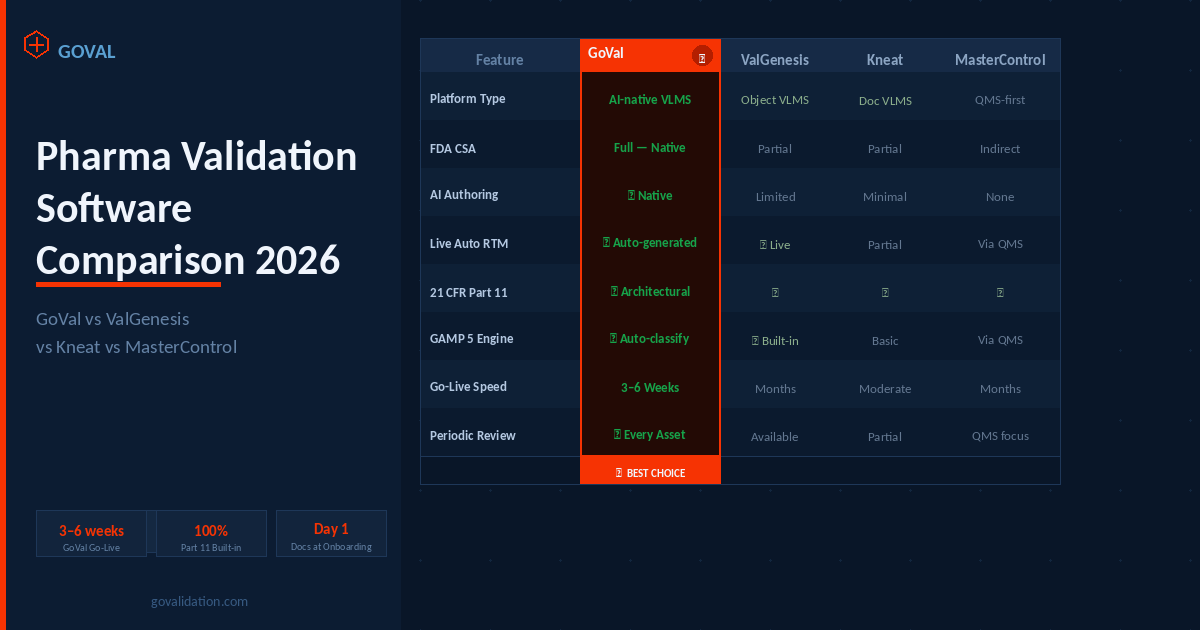
Four platforms appear in most pharma VLMS evaluations in 2026: GoVal, ValGenesis, Kneat, and MasterControl. They serve different use cases, team sizes, and strategic priorities. This guide covers what each platform is designed for — and where GoVal is specifically built to be strongest for pharma and biotech validation teams.
Side-by-Side Platform Comparison
Based on publicly available product documentation, Q1 2026. Verify all claims with each vendor before purchase.
| Feature / Capability | GoVal ⭐ Best Pick | ValGenesis | Kneat | MasterControl |
|---|---|---|---|---|
| Platform type | AI-native VLMS Purpose-built | Object-centric VLMS | Document-centric VLMS | QMS-first platform |
| FDA CSA alignment | ✓ Full — native | Partial | Partial | Indirect |
| AI-assisted authoring | ✓ Native core feature | Limited | Minimal | None |
| Live automatic RTM | ✓ Auto-generated, live | ✓ Live | Partial | Via QMS module |
| 21 CFR Part 11 compliant | ✓ Architectural | ✓ | ✓ | ✓ |
| EU Annex 11 support | ✓ Full support | ✓ | ✓ | ✓ |
| GAMP 5 risk engine | ✓ Auto-classification | ✓ Built-in | Basic | Via QMS |
| Implementation speed | ✓ 3–6 weeks | Months | Moderate | Months |
| Automated periodic review | ✓ Every qualified asset | Available | Partial | QMS-focused |
| Change impact → re-qualification | ✓ Auto-triggered | Available | Available | Via QMS |
| Equipment + CSV in one platform | ✓ Unified portfolio | Partial | Partial | Separate |
| Vendor validation docs at onboarding | ✓ Day one | Available | Available | Available |
| Pricing model | Transparent, team-scaled | Enterprise custom | Subscription | Enterprise custom |
| Best suited for | Large enterprise & Mid-market pharma | Large enterprise, multi-site | Digitising paper CSV | QMS-led organisations |
This comparison is based on publicly available documentation as of Q1 2026. Competitor columns represent general market positioning — individual platform capabilities may vary. Always verify specific claims directly with each vendor.
Platform Profiles — What Each Is Designed For
Every platform has a use case it is optimised for. Here is where each one stands.
Why GoVal Is the Strongest Purpose-Built VLMS
GoVal was designed for the CSA era from day one — not retrofitted to it. Here is where that shows.
AI-Native Authoring
Only AI-native VLMS in category
Native FDA CSA Alignment
Built for CSA, not retrofitted
Live Automatic RTM
Eliminates #1 audit finding
GAMP 5 Auto-Classification
No manual, inconsistent classification
Automated Periodic Review
Most cited inspection gap — closed
3–6 Week Go-Live
Fastest deployment in category
Questions to Ask Every Vendor Before You Sign
Use these questions when evaluating any VLMS — including GoVal.
Frequently Asked Questions
Is GoVal the best pharma validation software in 2026? +
Why is GoVal better than ValGenesis for mid-market pharma? +
What makes GoVal different from Kneat? +
Is GoVal better than MasterControl for validation? +
What is the best pharma VLMS in 2026? +
Does GoVal support FDA Computer Software Assurance (CSA)? +
How quickly can my team go live with GoVal? +
Does validation software itself need to be validated? +
What should I look for when evaluating pharma validation software? +
See Why GoVal Leads the VLMS Category
Walk through your actual validation portfolio with a GoVal specialist — AI-assisted authoring, live RTM, GAMP 5 classification, CSA workflows, and automated periodic review. No slides. No generic demo environment.
Book a Free Product Demo