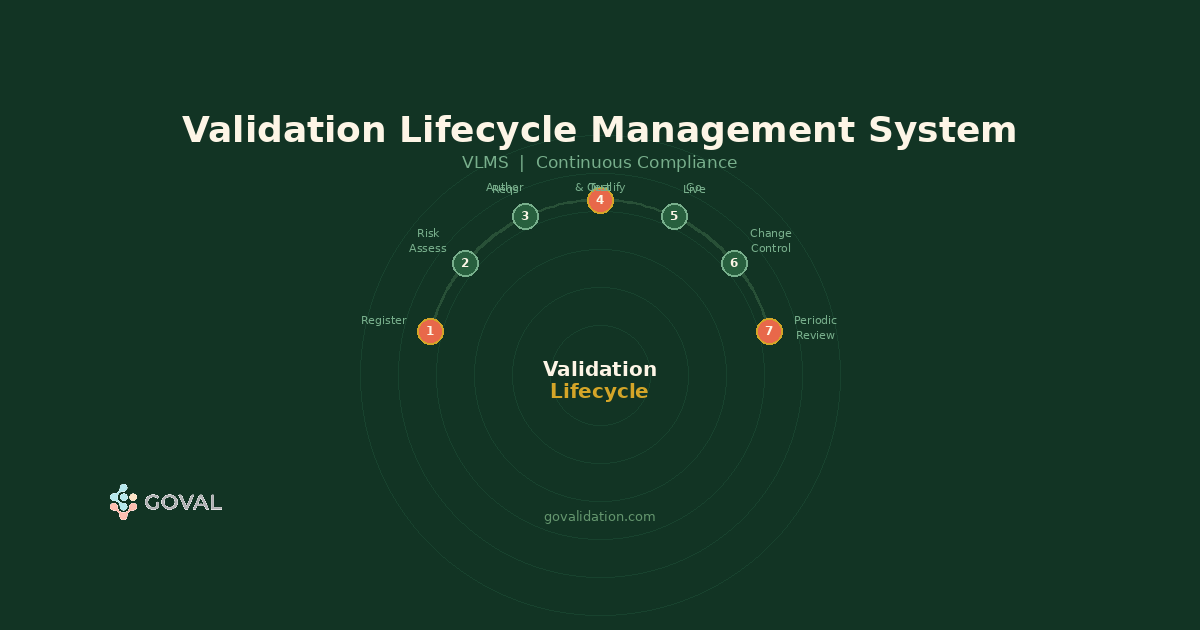
A Validation Lifecycle Management System (VLMS) maintains a continuous, current record of the validation state of every GxP computerised system in your environment — from initial registration through active validation, change management, periodic review, and retirement. Unlike project-based tools you use during qualification and then shelve, a VLMS is the ongoing system of record for your entire validated estate. When an inspector asks why a system is in a validated state right now, the VLMS is where that evidence lives — always current, never retrospectively assembled.
VLMS vs Document Management: The Critical Distinction
A document management system stores files. A VLMS stores structured, interconnected records you can query in real time.
The Seven Lifecycle Stages a VLMS Must Cover
A VLMS that covers only some of these stages forces manual handoffs at every gap — where unvalidated states develop silently.
The Real Cost of Managing Validation Without a VLMS
What the workaround ecosystem looks like — and where it fails under inspection pressure.
Periodic review spreadsheet 6 months out of date
The review tracking spreadsheet was not updated when a system owner changed. Three systems are overdue. Nobody knows until the inspector asks for the review schedule.
Change control never linked to validation
IT applied a vendor patch without a change control entry. The qualification record still shows the previous version. Equipment has been operating outside its validated state for weeks.
Deviation closed in QMS but not in qualification record
A test failure was corrected and closed in the QMS. Nobody linked the resolution back to the qualification record. The RTM still shows an open deviation against a validated requirement.
Audit prep takes a week instead of a click
Assembling a complete validation package from SharePoint folders, the QMS, the calibration system, and email approvals takes days. Each hour spent compiling is time the inspector is waiting.
What GoVal Delivers as a VLMS
GoVal covers all seven lifecycle stages in one connected platform — no manual handoffs, no silent gaps.
| What You Need | How GoVal Delivers It |
|---|---|
| Real-time portfolio visibility | ✓ Dashboard showing validation status, overdue reviews, and open deviations for every system — always current, no manual compilation |
| Automatic traceability (RTM) | ✓ Live RTM linking every requirement to risk assessment, test cases, and results — updated in real time throughout the lifecycle |
| Integrated change management | ✓ Changes logged against validated systems trigger automatic impact assessments and route re-qualification workflows where needed |
| AI-assisted authoring | ✓ AI assistance for URS drafting, risk assessment completion, and test case generation AI |
| Audit-ready at all times | ✓ Complete validation records and linked evidence available immediately — not assembled over days in response to an inspection notice |
| CSA and CSV in one platform | ✓ Risk-adaptive documentation scaling to each system's GAMP category — proportionate CSA evidence or full CSV documentation |
| Multi-framework support | ✓ 21 CFR Part 11, EU Annex 11, GAMP 5, and FDA CSA — all supported simultaneously for dual-regulated organisations |
Frequently Asked Questions
Questions from pharma validation teams evaluating VLMS platforms for the first time or switching from legacy systems.
What is a Validation Lifecycle Management System (VLMS)? +
What is the best VLMS software for pharma in 2026? +
How is a VLMS different from a document management system? +
How much does a VLMS cost for a mid-size pharma company? +
Which regulatory standards does a VLMS need to support? +
What is the ROI of implementing a VLMS? +
How many systems can a VLMS manage? +
What is the difference between a VLMS and CSV software? +
See Your Validated Estate in One View
GoVal gives you a real-time picture of your entire GxP portfolio — validation status, periodic review schedule, change history, open deviations, and linked qualification evidence — always current, always audit-ready.
See Your Validated Estate in GoVal