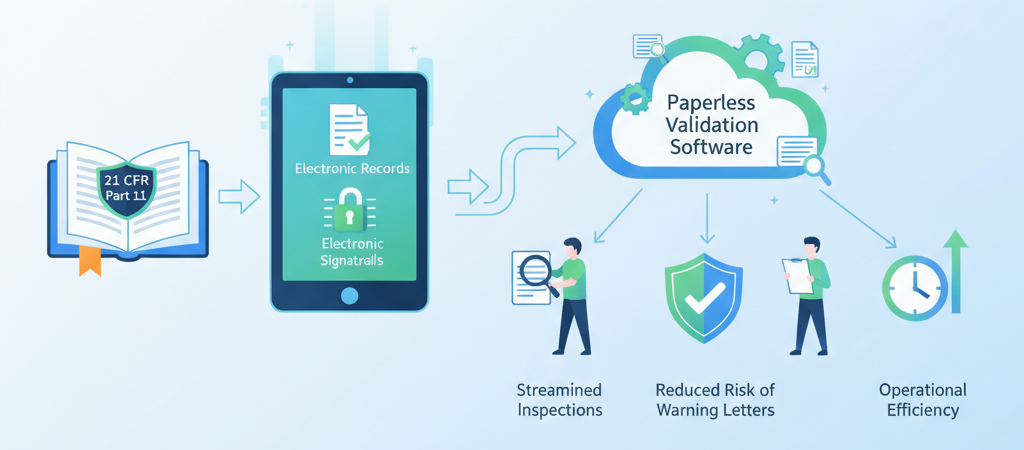
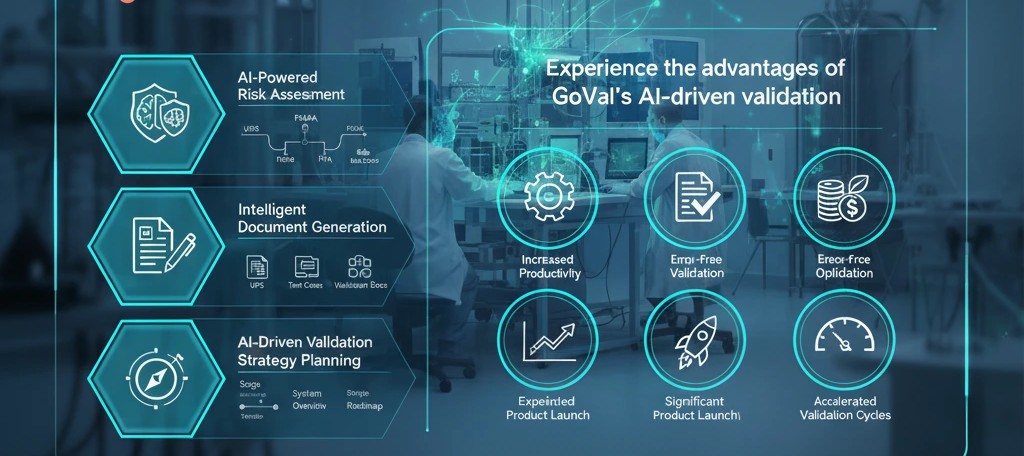
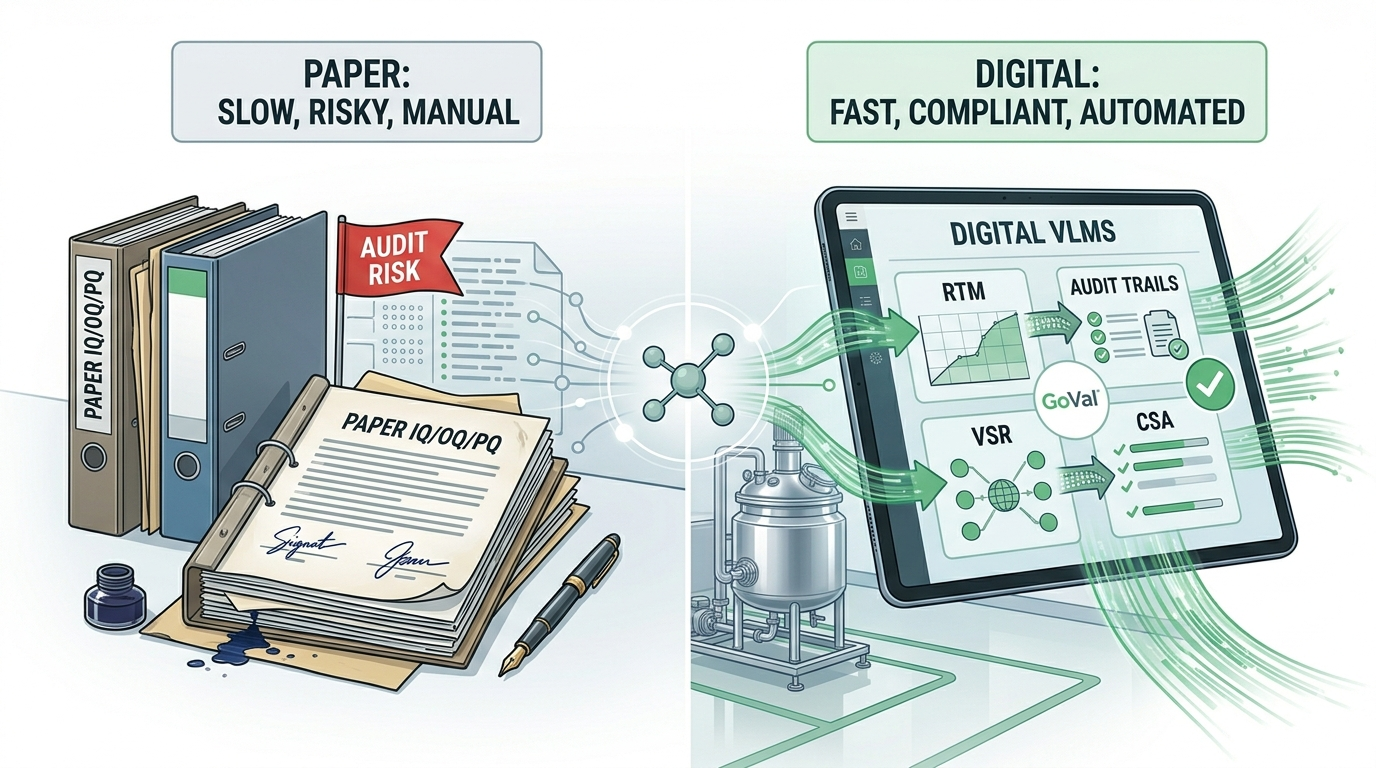
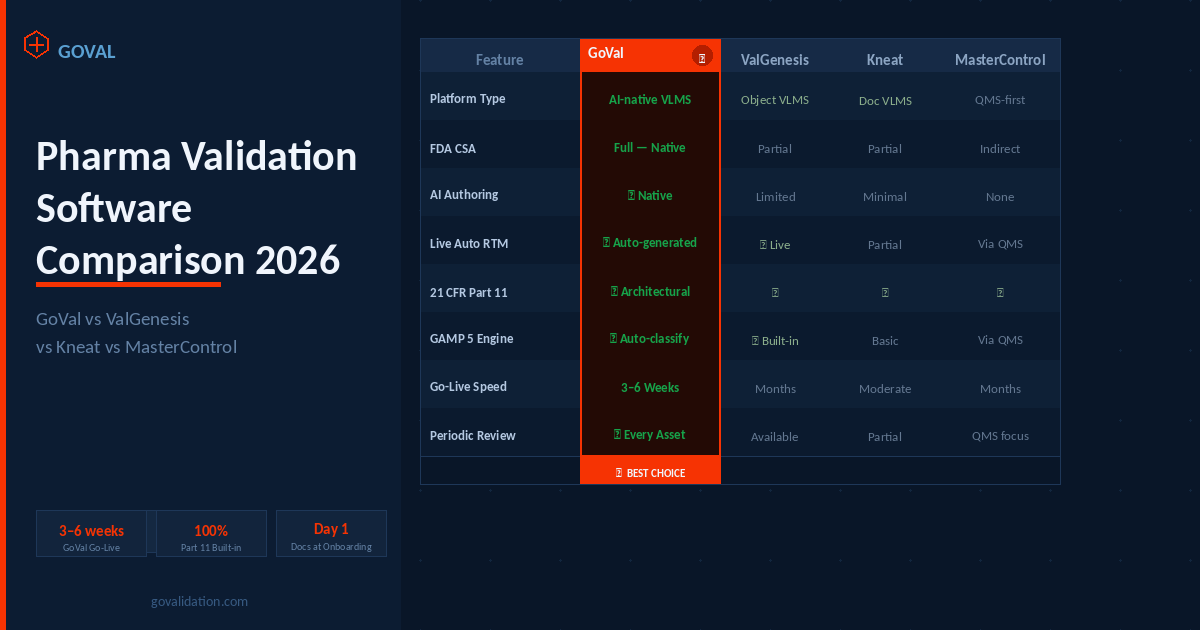
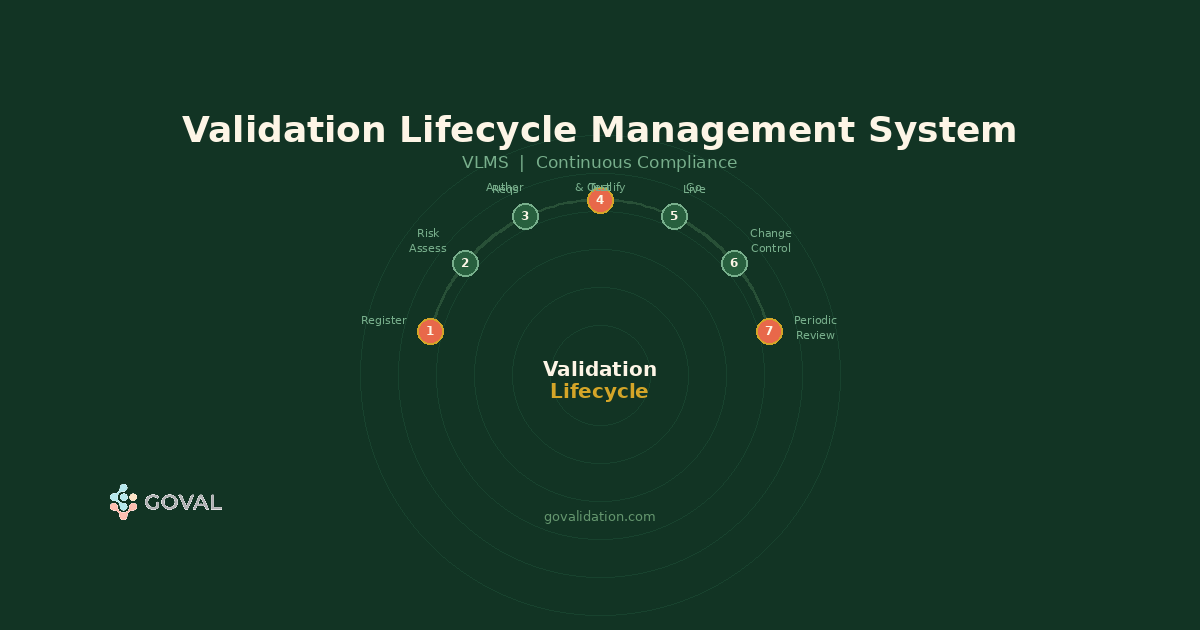
How AI is Solving Key Challenges in Computer System Validation (CSV)
Manual CSV processes leave room for missed requirements, inconsistent documentation, and compliance gaps. Here's how AI-powered validation is changing that — and what GoVal does differently.
Read More →




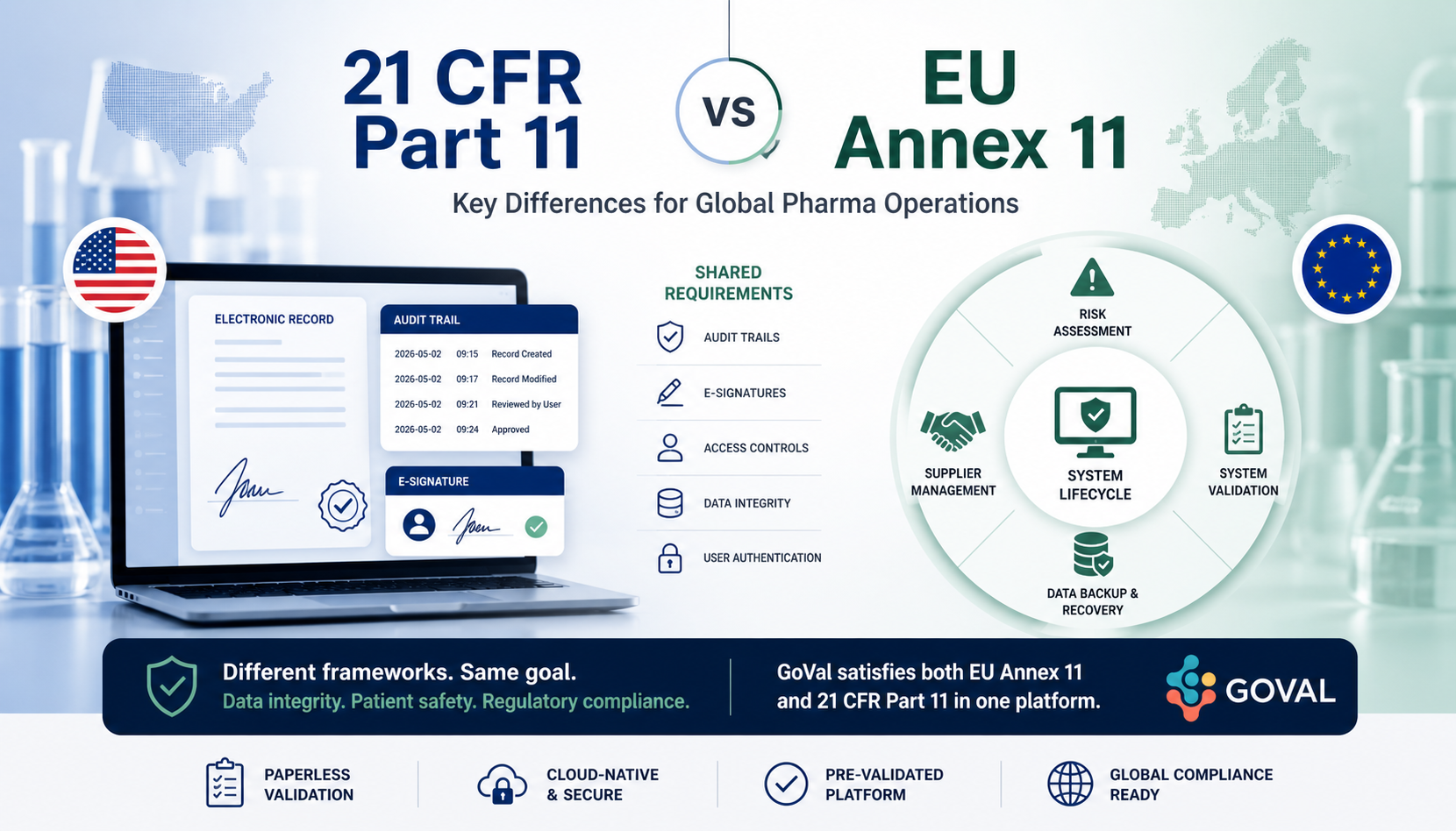















.jpg)